What Is a Rotary Evaporator? A Clear Guide for Lab and Production Users
This page explains what a rotary evaporator is, how it works, and how to choose the right model from 2 L to 50 L using real technical data.
I work every day with rotary evaporator users in chemistry, pharmaceuticals, plant extraction and food research. The goal here is simple: explain rotary evaporation in plain language and show exactly which problems it can solve.

Typical laboratory rotary evaporator system with water bath, condenser and vacuum connection.
1. What Is a Rotary Evaporator?
A rotary evaporator (often called “rotovap” or “rotary evap”) is a device used to remove solvents gently and efficiently from samples by evaporation. It combines three key elements:
Rotation of the flask to increase surface area
Controlled heating with a water or oil bath
Vacuum to lower the boiling point of the solvent
In simple words, a rotary evaporator is a controlled, safe, and efficient way to concentrate solutions, recover solvents, or dry samples without burning or degrading them.
According to data published in the journal Chemical Engineering & Processing, using reduced-pressure evaporation can cut energy consumption by more than 30% compared with open boiling at atmospheric pressure. A rotary evaporator is one of the most practical tools to achieve that in real labs.
2. What Problems Does a Rotary Evaporator Solve?
In daily laboratory and production work, several recurring problems appear:
Need to remove large amounts of solvent without degrading heat-sensitive compounds
Desire to recover expensive solvents (like acetonitrile, ethanol, methanol)
Long waiting time when evaporating samples in open flasks or on a hot plate
Risk of bumping, splashing, or overheating the sample
A rotary evaporator solves these by combining gentle heat, vacuum, and rotation to speed up evaporation while keeping the sample safe.
Why not just boil the solvent on a hot plate instead of using a rotary evaporator?
On a hot plate, the solvent boils at its normal boiling point and often bumps violently, causing loss of sample and risk of contamination. Under vacuum in a rotary evaporator, the boiling point drops significantly, so the solvent can evaporate at a much lower temperature. This protects heat‑sensitive compounds (for example, natural plant extracts or pharmaceutical intermediates) and improves solvent recovery efficiency.
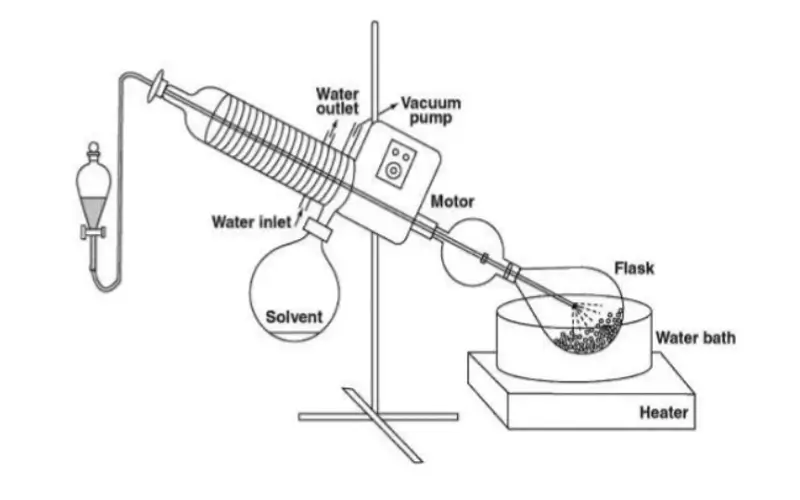
3. How Does a Rotary Evaporator Work? (Step by Step)
Although the instrument looks complex, the working principle is straightforward. A typical rotary evaporator system is composed of:
Rotating motor and evaporating flask
Heating bath (water or oil)
Condenser with cooling water or chiller
Receiving flask for condensed solvent
Vacuum pump and vacuum control
3.1 Basic working process
In operation, the process is usually:
Sample solution is poured into the round-bottom evaporating flask.
The flask is connected to the rotary evaporator and partially immersed in the heated bath.
The motor slowly rotates the flask (for example, 0–120 rpm), forming a thin solvent film on the glass surface.
Vacuum is applied, reducing the solvent boiling point.
The solvent evaporates, rises into the condenser, is cooled, and drips into the receiving flask.
The concentrated sample remains in the rotating flask.
Modern rotary evaporators, such as models with intelligent digital temperature control (for example, RT–399 °C with ±2 °C resolution in 2 L–5 L systems), allow precise control of the bath temperature and rotation speed, making the process repeatable and easy to scale.
For small-scale laboratory tasks, a 2 L rotary evaporator like the RE‑201D is often sufficient. It offers:
2 L evaporating flask and 1 L receiving flask
Stepless speed regulation from 0–120 rpm with a 40 W motor
Water/oil bath power 1200 W and temperature up to 399 °C
Evaporation rate > 1 L/h (water)
Ultimate vacuum < 133 Pa (about 1.33 mbar)
These parameters are enough for routine solvent removal in research laboratories.
4. Key Parameters to Understand on a Rotary Evaporator
When selecting a rotary evaporator, a few technical parameters directly affect performance and daily experience.
4.1 Evaporating flask volume
The evaporating flask size determines how much solution can be processed in one batch. Typical sizes include 2 L, 3 L, 5 L, 10 L, 20 L and 50 L.
| Model | Evaporating Flask (L) | Receiving Flask (L) | Rotating Speed (rpm) | Evaporation Rate (H2O) |
|---|---|---|---|---|
| RE‑201D | 2 | 1 | 0–120 | >1 L/h |
| RE‑501 | 5 | 3 | 0–120 | >1.5 L/h |
| RE‑1002 | 10 | 5 | 0–120 | >3 L/h |
| RE‑2002 | 20 | 10 | 0–90 | >5 L/h |
| RE‑5002 | 50 | 20 | 0–90 | >9 L/h |
A common rule of thumb is to fill the flask only to about one-half or two-thirds of its total volume to avoid bumping into the condenser. This means a 5 L rotary evaporator is comfortable for about 2–3 L of solution per run.
4.2 Bath temperature and control
For most organic solvents, water bath temperatures between 30–80 °C are enough. Many rotary evaporators support RT–99 °C and even up to 250 °C with oil, but in practice the actual setpoint depends on the boiling point under vacuum.
Intelligent digital controllers with ±1–2 °C stability keep the process consistent. According to multiple process optimization studies in pharmaceutical development, precise temperature and vacuum control can improve recovery yield of sensitive compounds by 10–20% compared with manual control alone.
Is a higher bath temperature always better for faster evaporation?
Not necessarily. Higher temperature can speed up evaporation, but it also raises the risk of thermal degradation and bumping. The most efficient point is usually “as low as possible while still giving a stable boil under vacuum.” That is why matching vacuum level and bath temperature to each solvent is more important than simply increasing temperature.
4.3 Rotation speed
Rotation speeds like 10–140 rpm (for example, in the R‑1005 to R‑1050 series) help spread the liquid into a thin film on the flask wall. Thin films evaporate faster because of the large surface area and constant renewal of the liquid surface.
Low-viscosity solvents can use higher speeds, while viscous extracts and foaming solutions may require lower speeds to keep the process stable.
4.4 Vacuum level
Ultimate vacuum is often specified around <133 Pa (1.33 mbar). In practice, the working vacuum is set higher than this to match the solvent. For example, ethanol may be evaporated at 35–45 °C with a vacuum of 80–150 mbar.
A stable vacuum is more important than the absolute deepest vacuum. Fluctuating vacuum can cause bumping and irregular boiling.
5. Common Types: From Small Lab Units to Large-Scale Systems
Rotary evaporators are available for many scales and use-cases. Understanding these categories helps avoid overspending or undersizing.
5.1 Small and mini rotary evaporators (1–2 L)
A small or mini rotary evaporator is ideal for:
Teaching laboratories and universities
R&D projects with small batch sizes
Developing new formulations or methods
These systems have compact footprints, moderate power, and are easy to install on a standard laboratory bench.
5.2 Medium lab rotary evaporators (3–5 L)
Models like the RE‑501 5 L rotary evaporator offer a good balance between capacity and space. A 5 L system with a 3 L receiving flask and 1500 W bath power is common in synthesis labs, pilot projects, and process development teams.
5.3 Large scale rotary evaporators (10–50 L)
For industrial extraction and pilot production, large systems like the RE‑1002, RE‑2002 and RE‑5002 are used as large scale rotary evaporators. Typical features include:
Evaporating flasks 10–50 L and receiving flasks 5–20 L
High-power baths from 3000 W up to 9000 W
Vertical double condensers or main cold + side cold high-efficiency condensers
Manual or electric lifting of the bath with 0–220 mm lift height
Brushless motors with 250 W and speeds of 0–90 rpm for stable operation
These systems can handle evaporation rates >9 L/h of water, which translates to even higher rates for solvents with lower boiling points.
Series such as R‑1005, R‑1010, R‑1020 and R‑1050 combine electric lifting, stainless steel baths, and vertical double-coil or reflux condensers. For example:
R‑1005: 5 L flask, 3 L receiving flask, 2 kW total power
R‑1010: 10 L flask, 5 L receiving flask, 3 kW total power
R‑1020: 20 L flask, 10 L receiving flask, 4 kW total power
R‑1050: 50 L flask, 20 L receiving flask, combined electric + hand lift

Large-scale rotary evaporator for plant extraction and pilot production applications.
6. How to Choose the Right Rotary Evaporator Size
When choosing a rotary evaporator, several practical questions should be answered upfront:
What is the typical volume of solution per batch?
How fast does the evaporation process need to be?
Are the compounds highly heat-sensitive or unstable?
Is there a plan to scale up in the near future?
Is it better to buy a larger rotary evaporator now to avoid upgrading later?
Not always. A 50 L system is powerful, but it requires more space, a stronger chiller, more solvent, and a higher initial investment. For many labs, a 5 L or 10 L system covers 80–90% of daily tasks with easier operation and lower running costs. The best choice is usually the smallest size that can comfortably handle the typical batch volume with some safety margin for growth.
As a rule:
Up to 1 L batches: consider a mini or 2 L rotary evaporator.
1–3 L batches: a 3–5 L rotary evaporator is usually ideal.
3–10 L batches: 10 L systems provide a good balance.
10–20 L per run: 20 L rotary evaporators are recommended.
More than 20 L per run: a 50 L large-scale rotary evaporator is suitable.
7. Typical Applications of Rotary Evaporators
Rotary evaporators are used across many industries:
Chemical synthesis: Removing solvents after reactions, concentrating reaction mixtures.
Pharmaceuticals: Concentrating active pharmaceutical ingredients (APIs), recovering chromatographic solvents.
Natural product extraction: Concentrating plant extracts, essential oils, and herbal preparations.
Food and flavor: Concentrating aromas and flavor compounds without damaging delicate components.
Academic research: Teaching distillation, solvent recovery, and sample preparation methods.
A study published in the journal Food Chemistry reported that using vacuum-assisted rotary evaporation for flavor extraction increased aroma compound recovery by up to 25% compared with atmospheric distillation, while better preserving heat-sensitive components. This illustrates why the technique is widely adopted in R&D labs.
8. Safety and Daily Operation Tips
A rotary evaporator is generally safe when operated correctly, but several habits can prevent accidents and downtime:
Increase vacuum gradually to avoid sudden boiling and bumping.
Use appropriate glassware sizes and never overfill the flask.
Check PTFE and fluoro rubber seals regularly for wear and replace if necessary.
Ensure cooling water or chiller is running before starting evaporation.
Do not set the bath temperature much higher than needed for the solvent.
Many modern systems include electric lifting, digital displays, and over‑temperature protection. For example, R‑series rotary evaporators use stainless steel water baths and electric lifting with well-defined lift height, making it easy to position the flask safely.
9. Summary: What a Rotary Evaporator Can Do for a Lab or Plant
A rotary evaporator is more than just glassware. It is a complete system designed to:
Remove and recover solvents efficiently under vacuum
Protect heat-sensitive compounds with gentle evaporation
Shorten processing time compared with open boiling or static evaporation
Scale from milliliter-level research up to tens of liters in pilot or production
Whether the need is a small laboratory rotary evaporator for daily sample preparation or a 50 L large-scale rotary evaporator for industrial extraction, the core principle remains the same: combine rotation, heat and vacuum to achieve controlled, efficient evaporation.
With the technical parameters and practical selection guidance above, it becomes much easier to choose the right rotary evaporator and use it safely and effectively in real projects.
Note: The performance figures and parameter ranges mentioned here are based on typical specifications of RE‑ and R‑series rotary evaporators and peer‑reviewed process studies. Actual values depend on configuration, operating conditions and solvent properties.

E-mail:
WhatsApp:
Address:
19/F, Block B, Guohong Mansion, Hi-Tech Development Zone, Zhengzhou City, Henan Province, China
Related blogs
You May also like
How Do Rotary Evaporators Work? A Practical, Easy Guide for Faster Solvent Removal
Learn how rotary evaporators work in plain English: vacuum + gentle heating + rotation + condensation. See key specs (2L–50L) and how to choose the right rota...
Read MoreRotovap Distillation | Complete Guide to Rotary Evaporators
Learn everything about rotovap distillation, how rotary evaporators work, applications in chemical labs, and why they are essential for efficient solvent remo...
Read MoreComplete Guide to 50L Rotary Evaporators
Complete guide to 50 liter rotary evaporators - working principles, applications, and industrial significance...
Read More